The unique structure of TCR
T-Cell receptor (TCR) is the key mediator in adaptive cell mediated immune response. A complex molecular mechanism, many processes of which aren’t yet fully known, underlies the intrinsic ability of TCRs to recognize a broad range of their target molecules. These target molecules or binding partners of TCRs are universally linear antigenic peptides which bind to major histocompatibility complex molecules (p-MHC) arrayed on antigen presenting cells (APCs). In fact, APCs should first bind to the antigenic peptides in order to offer them to TCRs. Therefore, at the heart of the immune response process is a receptor-ligand interaction between variable domains on the TCR and a peptide cradled in the groove of MHC molecules.
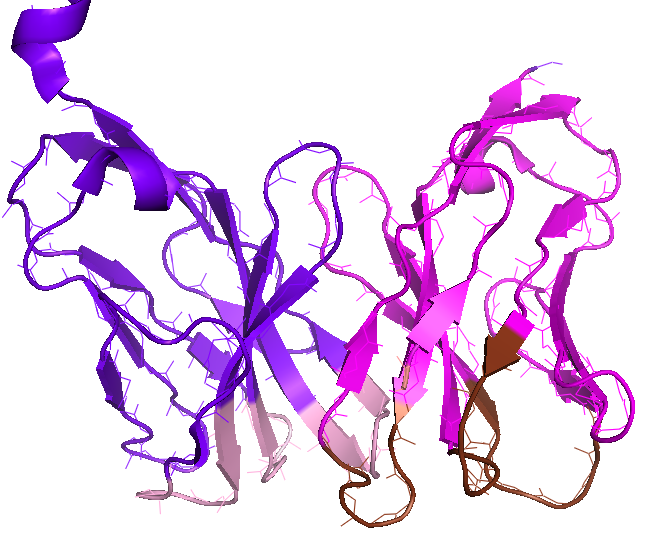
The majority of T-cells express TCR as a heterodimer protein consisting of alpha- and beta-chain; Each chain in TCR possesses two specific domains; While C-domain is required for the complete localization of TCR on the cell surface, V-domain from gene segments – V(variable) and J(joining) genes recombination for alpha-chain (colored in magenta in figure) and V, J, and D(diverse) genes recombination for beta-chain (colored in purple) – serves as antigen recognition site. This subpart of TCR, shown in figure (PDB 3TF7), interacts with p-MHC complex. The site-specific gene-recombination in V-domain of the TCR generates the extraordinary ligand specificity and sensitivity of TCRs. Six loops, three for alpha-chain (brown) and three for beta-chain (pink) in V-domain, denoted as complementarity determining region (CDR), determine the specificity of TCR for p-MHC ligand.
MHC presenting platform for antigenic peptides
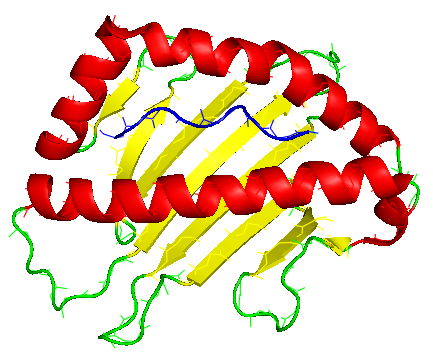
To ensure presenting a wide range of peptides to T-cells, MHC molecules, also called human leukocyte antigens or HLA in humans, are extremely polymorphic. Two main classes of MHC molecules are mainly different in the geometry of their antigen binding cleft: While MHC-I class antigen binding cleft is closed at the N-terminal and C-terminal regions, MHC-II molecules have an open-ended cleft. The preferred length of the bound peptide is defined by these differences between open and closed ends of the cleft; shorter peptides with 8-10 residues bind MHC-I and longer peptides with more than 11 residues are best matched with the open-ended cleft of MHC-II molecules.
The antigenic peptide (shown in blue) is first bound in the groove on the surface of MHC protein. A large 8-stranded ß-sheet (shown in yellow) forms the bottom of the antigen-binding groove of the MHC molecule. This ß-sheet is a common feature of every MHC molecule. With two flanking helices exposed for interaction (shown in red), MHC molecule offers the linear antigenic peptide to TCRs creating a relatively flat TCR-pMHC contacting face. The ß-sheet floor enforces a common TCR diagonal docking onto MHC.
Structural geometry of TCR-pMHC binding
The most common binding topology of TCR-pMHC is a diagonal binding mode in which the V_beta domain of TCR contacts the Alpha_1 helix of MHC and V_alpha domain of TCR contacts the Alpha_2 helix of MHC molecule. Any alternative binding topology might still happen between two molecules but with a more dramatic twist and/or shift of TCR respect to pMHC. These unconventional docking modes might not lead to productive T-Cell activation. In fact, TCR can best achieve contacts with the antigenic peptide in MHC groove via a diagonal docking geometry.

Most significant contributors to MHC from TCR are CDR loops. It is generally accepted that CDR3 loops, from both alpha- and beta-chain, mainly drive peptide recognition (left figure, middle section). CDR1 and CDR2 loops, on the other hand, are mainly focused on the recognition of MHC molecule itself, as shown in the left-side figure. The peptide specificity of TCR, therefore, is particularly defined by CDR3 loops.
The unique structure of TCRs as well as its diversity resulting from the somatic rearrangements of variable gene segments offer an exquisite specificity and sensitivity to T-cells for immunological recognition. But it is also the unique presentation of antigenic peptides through MHC molecules that enables the cross-reactivity of TCRs through a conventional and productive diagonal binding geometry.
References:
[1] “TCR Recognition of Peptide–MHC-I: Rule Makers and Breakers”, Christopher Szeto, et al.
[2] “T cell receptor signaling is limited by docking geometry to peptide-Major Histocompatibility Complex”, Jarrett J. ADAMS, et al,